Medical Device Label Compliance
Medical Device Label Compliance
Label compliance for medical devices involves adhering to regulatory standards and guidelines set by CDSCO (Central Drug Standards and Control Organization) governing the information presented on medical device labels.
Labeling compliance for medical devices is a critical aspect to ensure the safe and effective use of medical devices as precise labeling delivers essential information that helps prevent misuse and minimizes the risk of adverse events. Non-compliance with labeling regulations can lead to regulatory penalties, product recalls, and reputational damage, ultimately compromising patient safety and market access.
Under the Medical Devices Rules (MDR), 2017, issued by the Central Drugs Standard Control Organization (CDSCO), medical device labeling must meet specific requirements to ensure transparency, traceability, and user safety.
Key labeling requirements under MDR, 2017 include:
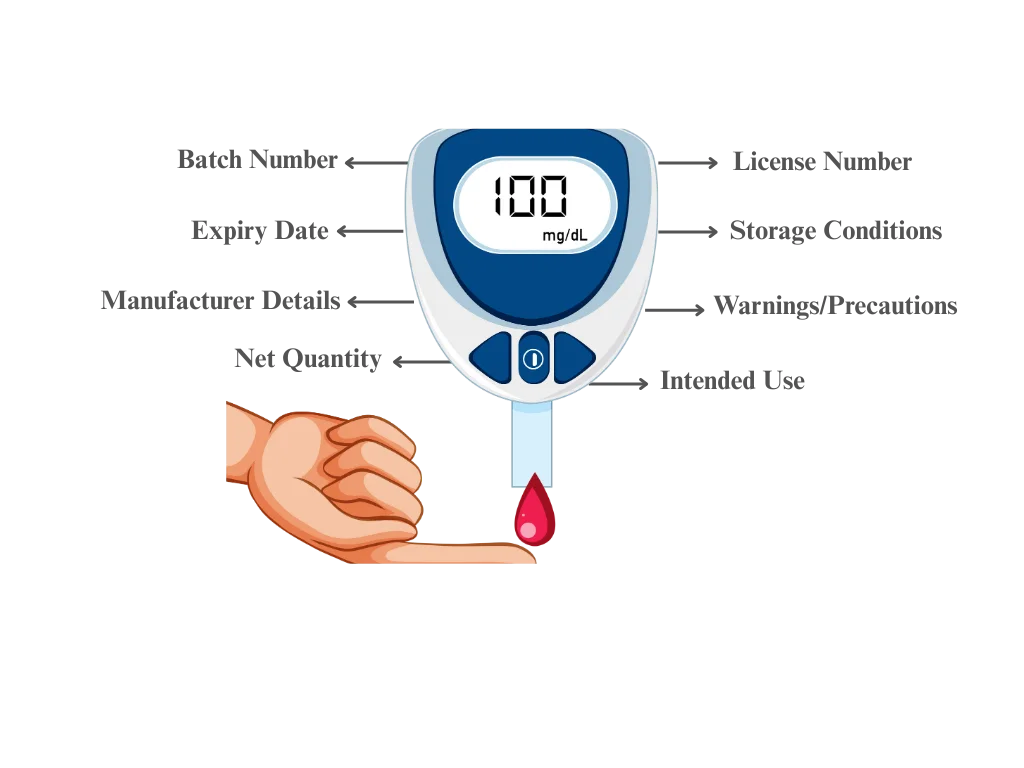
- Device name and intended use
- Manufacturer details
- Batch or lot number
- Date of manufacture and expiry
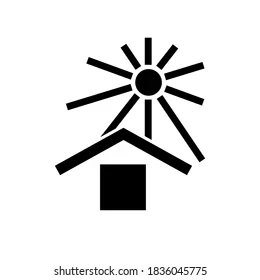
- Storage conditions
Compliance-Driven Labelling: Benefits Beyond the Basics
At Move Ahead, we help ensure your medical device labels are fully compliant with CDSCO’s MDR 2017 requirements—enabling smooth regulatory approvals, risk-free market entry and sustained compliance.
Use of ISO Symbols in Medical Device Labeling
ISO symbols play a vital role in medical device labeling by conveying important information in a clear, standardized, and universally understood format. These symbols help users quickly identify essential details such as handling instructions, storage conditions, sterility, and safety warnings—without language barriers. As per ISO and the Medical Devices Rules (MDR), 2017, the inclusion of appropriate ISO-compliant symbols is crucial to ensure global regulatory acceptance, enhance user safety, and maintain labeling

Do not use if package is damaged and consult instructions for use
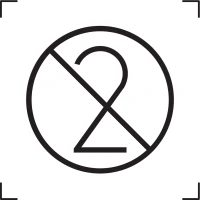
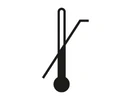
Indicates the date after which the medical device is not to be used.

In Vitro Diagnostic Medical Device
Should only be used to identify IVDs and note that the device is for “in vitro use”
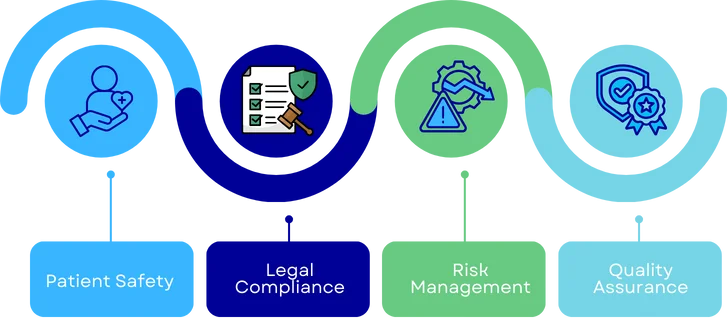
Benefits of Labelling Compliance
- Patient Safety - Ensures users have accurate and essential information for the device's safe and effective use, minimizing the risk of misuse or adverse events.
- Legal Compliance - Meets the mandatory regulatory requirements of bodies like the FDA and MDR, preventing legal issues.
- Risk Management- Minimizes potential product misuse and liability by providing clear and compliant information.
- Quality Assurance- Reflects adherence to industry standards and reinforces trust in the product’s safety and performance.
How MAC can help you?
Move Ahead Consultancy specializes in ensuring your medical device labeling complies with Indian MDR, 2017 requirements. Our experts meticulously review and validate all label content including artwork, symbols and mandatory details for accuracy and regulatory alignment. We simplify complex global standards and help you significantly reduce compliance risks, establish trust and maintain uncompromising patient safety.
Captcha:

Move Ahead at a Glance
Years of Experience
Employees
Happy Clients
Projects
Our Clients